 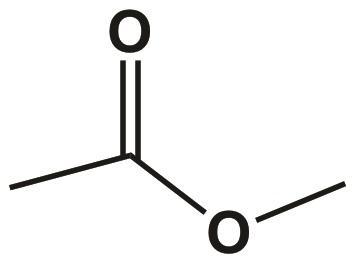
Hydrogen bonding between C-H groups and oxygens, although possible in principle (see comments to your question) and, for example, found in protein structures, can be neglected in this case due to the low number of donors and the weakness of the interaction (< 17 kJ/mol). The permanent dipoles can also interact with each other, in the form of intermolecular attractive interactions between centers with $\delta $ and $\delta-$ partial charge (and likewise repulsion between centers of equal partial charge). They are responsible for the weak interactions between the alkyl chains, like in other alkanes and non-polar molecules. London dispersion forces, which result from short-lived dipoles induced by fluctuations in the electron shell of molecules, are also present. As both dipole moment vectors point roughly in the same direction, they do not cancel each other out like in the case of carbon dioxide, so the molecule has a net dipole moment and is therefore (weakly) polar. It does not store any personal data.As you have already figured out, diethyl ether is a small dipole due to the electronegativity difference between carbon and oxygen, which can also be seen in this electron density plot. The cookie is set by the GDPR Cookie Consent plugin and is used to store whether or not user has consented to the use of cookies. The cookie is used to store the user consent for the cookies in the category "Performance". This cookie is set by GDPR Cookie Consent plugin. The cookie is used to store the user consent for the cookies in the category "Other. The cookies is used to store the user consent for the cookies in the category "Necessary". 
The cookie is set by GDPR cookie consent to record the user consent for the cookies in the category "Functional". The cookie is used to store the user consent for the cookies in the category "Analytics". These cookies ensure basic functionalities and security features of the website, anonymously. Necessary cookies are absolutely essential for the website to function properly. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
